This book is designed to both update the reader on the
status of the Beilstein database and search system and derived electronic
information, as well as provide the chemistry community with a
comprehensive and up-to-date view of the overall activities and
scope of the current Beilstein system. When the first ACS book on
Beilstein was published in 1990
(1) there was a
renewed and growing interest in Beilstein, as it moved from its
past environment of a print based German language reference, existing
only as a very large series of books (which made up the
Beilstein Handbuch der Organischen Chemie - or, in English,
the Beilstein Handbook of Organic Chemistry). In the short
time since 1990, the staff of Beilstein (first in the Beilstein Institute
and now in Beilstein Information Systems) have continued
their vast modernization program, which has resulted in many new chemistry
data and information resources for the chemical community. This
book is a tribute to Konrad Beilstein,
(Figure 1), the man who
had the foresight to realize that high quality scientific
chemical data and information would be a timeless resource for
chemists.

Throughout the history of abstracting scientific
literature in chemistry, the name Beilstein has had a unique position
of quality and value to the chemist. As a young graduate student
in organic chemistry I first came across Beilstein as part of
my synthesis work on a bicyclic nitrogen ring system. In
1963 it took me a considerable amount of time to search for this
class of compounds and translate the appropriate sections of
the Beilstein Handbook into English; now those activities take a few
seconds, because
the database is computerized and the information is essentially
all in English. The transformation of this sleeping giant into
a modern information resource is amazing and of great
value to the ongoing progress of most of academic and industrial
chemistry.
Table 1.1 is a brief chronology of Beilstein - the man, the Handbook,
and the database. It is easy to see that the period between the
first ACS book and this book was a time of major development and
change at Beilstein.
Introduction to the Beilstein System
1838: Friedrich Konrad Beilstein was born in St. Petersburg, Russia
1865: Beilstein became Professor of Organic Chemistry, St. Petersburg
1866: Beilstein became the Chair of Chemistry at the Imperial Technical Institute in St. Petersburg
1881: First Edition of the Beilstein Handbook (2 volumes, 1500 compounds, 2200 pages)
1885: Second Edition (3 volumes, 4080 pages)
1906: Third Edition (8 volumes, 11,000 pages)
1906: Death of Friedrich Konrad Beilstein
1918: Fourth Edition (German Chemical Society, Berlin)
1984: Fifth Edition in English (480 volumes, 400,000 pages)
1988: Beilstein Online on STN
1989: Beilstein Online on DIALOG
1990: ACS Symposium Series book - The Beilstein Database
1995: CrossFire - Beilstein file in-house with ca. 6,000,000 organic compounds with properties data
1996: CrossFire Gmelin - Gmelin file in-house available with ca. 1,000,000 inorganic and organometallic compounds with properties data
1997: ACS Book - The Beilstein Database and Search
System
Even though the Beilstein Handbook of Organic Chemistry
covers Aonly@ organic chemistry, it has been a critical resource for most of
the 20th century. As we approach the 21st century the management
and leadership of Beilstein Information Systems GmbH has further
developed this valuable resource into a number of practical, every-day,
tools for the chemist. The purpose of this book, the successor
to the ACS Symposium Series book entitled "The
Beilstein Online Database - Implementation, Content, and Retrieval",
is to demonstrate how the Beilstein database and associated software
products have been evolving over the last 7 years since the
first book was published.
With all the changes, remodeling, and reorganization
of Beilstein in the past few years into two separate organizations, the
Beilstein Institute
and Beilstein Information Systems (explained in further detail
later in this chapter) it is probably best
to first answer the question - "What
is Beilstein, now?" Perhaps the best way to begin to answer
this question is to show what Beilstein was in the past. Beilstein
is an enormous set of reference books
(Figure 1.2) that, although
very well organized, occupied a great deal of space. It was
relatively difficult to quickly locate information spread throughout
the five different series composing the handbook.

Figure 1.2. The current Beilstein Handbook, from
the Basic Series to Supplements I - V.
To find out
what Beilstein is today, one need only
search a number of Internet resources. A recent search
showed many citations of the word "Beilstein".
For example, the Lycos
(2) search engine retrieved almost
600 records. Other search engines, such as, Alta-Vista
(3) produced some
200 citations, whereas InfoSeek
(4) produced 76 citations, and WebCrawler
(5) produced 59 citations. Lastly Yahoo!
(6) found Beilstein
in just one citation, the Beilstein web site. These various citations
included a number of references to the Beilstein Internet World
Wide Web (WWW) site,
www.beilstein.com as well as many chemistry
department www sites around the world that
have either the Beilstein Handbook or access to Beilstein in
electronic
format. There are also references to Beilstein the town in Germany,
Beilstein jade, the Beilstein mountain in Austria, and so on.
Although there are many answers to "What
is Beilstein, now?", this book will cover only those that
relate to the areas of chemistry for which the Beilstein Institute
(7)
and Beilstein Information Systems
(7) are involved. When
the first ACS book was published in 1990,
it was easy to say that Beilstein was the Handbook and the online
database. Today, a mere 7 years later, the name "Beilstein"
represents the Handbook, Beilstein Online, CrossFire, CrossFire
plus Reactions, and CrossFire Gmelin, Autonom, Current
Facts, and more.
Evolution of the Beilstein Institute
To create this "new world" of the 21st century Beilstein, a new administrative approach was developed and implemented by the two presidents of the Beilstein Institute during the early 1990's, Clemens Jochum and Reiner Luckenbach. The evolution of the Beilstein Institute from a world class institute to a world class modern organization, involved a considerable change in approach, attitude, and internal and external operations. Beilstein has now evolved and remade itself into a commercial venture, and it is run as a business, in a most business-like manner. The transformation of all staffing and financial activities, from outsourcing the creation of the database, to the termination of German government support, to having all services paid for by users - a critical goal for the success of the Beilstein system - has produced a completely new Beilstein. Virtually nothing but the name and high quality are the same after this massive reorganization effort.
Beilstein
has also changed administratively from being only the Beilstein
Institute to being both the Beilstein Institute and Beilstein
Information Systems. Beilstein, which was funded for years by
the German publisher Springer-Verlag and the German government,
is now changed and a private company, Information Handling
Systems (IHS), is responsible for the finances. A new corporate
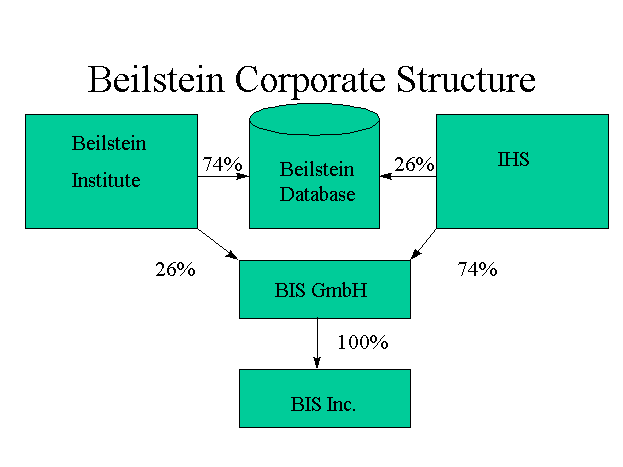
structure, shown in
Figure 1.3
(8), shows these overall relationships.
In this new structure, the database is now 74% owned by the
Beilstein Institute and 26% owned by IHS. The marketing company,
Beilstein Information Systems GmbH, is 74% owned by IHS and 26% owned
by the Beilstein Institute. Lastly, the American subsidiary, Beilstein
Information Systems Inc, is 100% owned by the German company Beilstein
Information Systems GmbH.
The new Beilstein database is now marketed and sold by IHS, a
highly successful information company, which chose Beilstein as
its first project in the scientific electronic database area.
The Beilstein Institute staff now only continues to produce
the Beilstein Handbook.
Lastly, software such as Autonom (the automatic structure
- nomenclature program) has been developed. It is from all of
these changes and additions to Beilstein that the title of this
chapter, The Beilstein System, was chosen. Because it is now
a total system is made of many parts and activities, it
seems to be the most appropriate term to use.
Chemical Abstracts and Beilstein
One other point needs to be stressed before going further. The Beilstein system is primarily a system of data, which is extracted and processed information. Chemical Abstracts Service, a part of the ACS, has produced Chemical Abstracts (CA) since 1907. CA is another valuable and critical source of chemical information. Chemical Abstracts and Beilstein are often thought of as competitors by some in the field of chemical information. In reality, the CA and Beilstein databases are complementary tools. CA covers virtually all of the chemical literature - organic, inorganic, physical, analytical, polymer, materials, and so on. CA also covers the literature primarily from a very different and valuable perspective by providing an abstract and a summary that is a synopsis of each article. In comparison, Beilstein covers primarily the chemical literature focused on organic chemistry dating back as far as 1771. Beilstein covers the chemical literature in which scientific data is presented, providing all the data presented in each published article as given by the original authors. Even though Beilstein has covered "classical" chemistry for most of the 19th and 20th centuries, as it moves into the 21st century it has responded to the needs of the chemical community and expanded its coverage of the literature to include toxicological and physiological effects of chemicals. Both CA and Beilstein have their audiences, which can overlap, but are often quite different and the applications of these two resources are also quite different. To obtain complete information, the research chemist must search both files to find all the, hard data and other information on any compound of interest.
NetFire
Two potentially important topics are covered in a very cursory way in this book, because they are too new for proper presentation and discussion. The first is NetFire, the new Internet based current awareness organic chemistry literature search service from Beilstein. Released in December 1996, and made available to the chemical community for testing and evaluation at no cost for the first half of 1997, there is insufficient experience with NetFire to include it here. There has been one short description of NetFire written (9). Basically NetFire is a database of journal titles, authors, and abstracts from the organic chemical literature from 1980 to the present. The NetFire database contains the titles, abstracts and authors of journal articles published in over 140 of the top journals in organic and medicinal chemistry. The Internet WWW query-by-form mechanism permits the formulation of a great variety of inquiries. You may search by author, by words included in the abstractor , by title (or words therein). You may also restrict your search to a certain journal, or time range. Also included in the output is a cross reference number to CrossFire, for those who have access to the entire Beilstein database.
CrossFire Gmelin System
The second topic not covered in this book is the CrossFire Gmelin system - which is the Gmelin database provided in-house under the Beilstein CrossFire software - The Gmelin database, produced by the Gmelin Institute of Frankfurt, Germany, is the most comprehensive collection of factual data on organometallic (coordination) compounds, alloys, glasses, ceramics, minerals and physical chemistry. At present the Gmelin database contains more than 1 million chemical substances and over 900,000 reactions reported in the literature from 1772 up to the present and Gmelin handbook citations from 1772 to 1975. Gmelin contents includes approximately:ca. 470.000 coordination compounds
ca. 55.000 alloys
ca. 14.000 glasses and ceramics
ca. 3.200 minerals
With the Gmelin database as part of the in-house
Beilstein CrossFire system, the two most comprehensive factual
databases covering the areas of organic, inorganic and organometallic
compounds are now readily available under a speedy, convenient
and user friendly client-server based software system.
Coverage of the Periodic Table
Before outlining the details of this book it is constructive to describe Beilstein's and Gmelin 's coverage of the periodic table of the chemical elements. In Figures 1.4 and 1.5 the chemical elements included in Beilstein and Gmelin are presented.
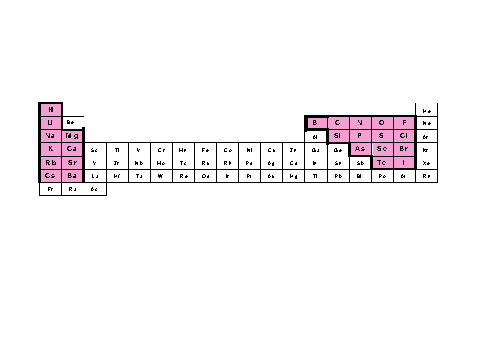
Figure 4 . The Periodic Table showing which elements
are covered in the Beilstein database
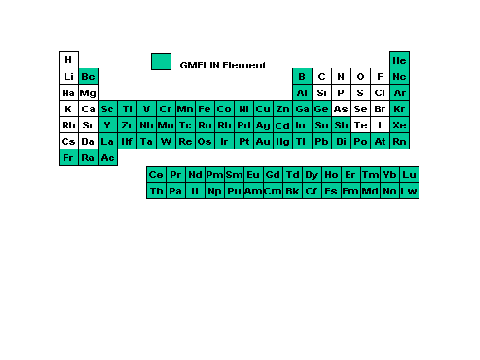 Figure 5. The Periodic Table
showing which elements are covered in the Gmelin database.
Figure 5. The Periodic Table
showing which elements are covered in the Gmelin database.
Chapter Overviews
Chapter 3
Even though the
Beilstein system has many new features, which are described in later
chapters of
this book, the Beilstein Online database, one of the original components
of the Beilstein system, is still a very valuable resource for
chemists.
Andreas Barth, of STN - Karlsruhe, who worked with
the Beilstein Institute in the late 1980's to develop the first
version of the online database has revised and updated his original
contribution from the ACS Symposium Series book. This chapter
includes information on the general structure of the information
in the database and in each chemical compound record. A variety
of search examples for data, physical properties, chemical reactions
and other information are given. Thus chapter 3 serves as the
basic reference point for most of the information contained in
this book.
Chapter 4
Wendy Warr , a well known
expert consultant in
chemical information, working with
Bernd Wollny at Beilstein Information
Systems describes the Current Facts database, its content,
search capabilities, and its value to the chemist. Current Facts
on CD-ROM is the Beilstein database of structures, data, and literature
citations, designed to be used on a PC to provide
recent information on chemicals reported in the literature. Current
Facts contains the 1 year's worth of information, updated quarterly,
with the most recent 3 months of information replacing the oldest
33 months. The database goes back to 1990. One nice
feature of this product is the structure searching capability
built into it.
Chapter 5
Chapter 5 begins the treatment of the new Beilstein for the
21st century. This chapter, "Computer Systems
for Substructure Searching", was written by one of the designers and
developers of CrossFire,
Dirk Walkowiak, of Softron GmbH, in
conjunction with
John Barnard, a chemical information consultant
and an expert in structure handling and search systems. The chapter
starts with a history and introduction to chemical structure searching.
From there the specifics of the CrossFire structure search system
(general architecture, search algorithms, structure coding, and
reaction retrieval) for the in-house Beilstein system is described
in this chapter for the first time. This chapter describes
the way in which CrossFire, which currently runs on the IBM
RISC System/6000 computer system with a UNIX operating system
(other platforms, such as DEC and Windows NT, will be available
in the near future) allows for very rapid searching of the more
than 7 million chemical structures in the Beilstein database.
Chapter 6
After the treatment of structure searching and CrossFire in Chapter 5, Alexander (Sandy) Lawson of Beilstein Information Systems, discusses CrossFireplusReactions in Chapter 6. Another improvement in the Beilstein system has been the extraction of reaction information which is the heart of the Beilstein Handbook of Organic Chemistry. One of the critical and unique features of Beilstein has been the fact that virtually all data reported in the chemical and related literature for a chemical substance are entered into the system. All compounds entered into the Beilstein database have actually been synthesized and the structure has been assigned unambiguously.
This chemical approach of having a database comprising
actually synthesized chemical substances (as opposed to the abstracting
approach of CA is one of basic features
of Beilstein that makes it so valuable to the bench chemist. In
CA, published papers are abstracted on the basis
of their being published in the chemical literature that CA
covers (some 13,000 journals) plus patents. Even though the
bulk of this material is the same information as is found in both
the Beilstein Handbook of Organic Chemistry and database,
there are differences. CA is a much larger database
because its coverage goes beyond organic compounds and includes
inorganic chemicals. Some chemicals that do not exist are in
the CA database for two main reasons. Either they are
chemicals that are being studied in theory or in computer analysis
and calculation programs, or they are needed by the CA indexing
system to allow for the proper indexing of derivatives (e.g.,
salts and parent ring systems) of a chemical. (This indexing
should not be viewed as negative, because the CA indexing system and
index database is one of its hidden virtues and a very valuable
resource.) CA, owing to its broader coverage
of the chemical literature, also has more reaction information
in many cases for chemicals from more recent publications.
The combination of the reaction information content
of Beilstein, which goes back to the 18th century,
and the powerful new Beilstein Commander makes the new Beilstein
product, CrossFireplusReactions, a valuable and unique
resource. CrossFireplusReactions is a database and search
system that should be a part of every synthetic organic chemistry
research laboratory.
Chapters 7 and 8
Even though the chemical industry is the major user of
the various Beilstein products, the chemist's first exposure to
Beilstein comes when he or she is in school, either as an undergraduate
or graduate student. Chapters 7 and
8, written by chemical information and library experts from leading
academic institutions in the United States and Europe,
Englebert (Bert)
Zass (Chapter 7) and
Ken Rouse
and
Roger Beckman (Chapter 8),
describe how Beilstein is being taught and used in the university
setting both in Europe and the United States.
In Chapter 7 by
Bert Zass (ETH, Switzerland) provides
an excellent comparison and discussion of a number of reaction databases
that are available both online and in-house, with emphasis
on the Beilstein reaction database and the Beilstein CrossFireplusReactions
system.
In Chapter 8,
Ken Rouse (University of Wisconsin)
and
Roger Beckman (Indiana University) describe a consortium of
universities that have joined forces to make the Beilstein database
available to a large community of students and academics via the
inhouse CrossFire system. In addition to discussing the very positive
response from the academic user community, the authors talk
about the economics and finances of the system.
Chapter 9
From this background of the CrossFire system and
examples of use in academia,
Wendy Warr,
in Chapter 9 discusses and explains its practical everyday value to the
industrial
chemist, and in particular to the pharmaceutical chemist. Training,
pricing, integration of information sources, NetFire, and CrossFire
Gmelin are all covered in this chapter.
Chapter 10 describes
Autonom, a software program that gives the chemical names of structures
followin International Union of Pure and Applied Chemistry (IUPAC)
nomenclature rules. This chapter, written by the developer of AUTONOM,
Janusz Wisniewski of Beilstein Information Systems,
is an detailed description of the performance of this program. The
algorithmic approach that was used is also discussed in detail.
AUTONOM, which stands for AUTOmatic NOMenclature, is a program
every chemist who has ever published a manuscript with a chemical
structure will love. Chemists can easily draw structures, but
few can name a structure according to the extensive and often
complicated IUPAC or ACS/CAS naming rules. Instead of referring
to structures as I, II, III, ... , XL, - AUTONOM gives IUPAC names
for organic and inorganic chemical structures. For companies
that need to register substances under their proper chemical names in
to obtain approval from government and regulatory
bodies, AUTONOM is a wonderful tool. This chapter describes version
2.0 of AUTONOM, the classes of organic and inorganic compounds
it covers, and its limitations.
By the time you reach the end of Chapter 10, you
will be both well versed in what Beilstein is today and is evolving
into in the future, as well as knowledgeable about how the various
Beilstein products can be of invaluable assistance to the everyday
activities of almost every chemist.
1. Heller, S. R., Editor, The Beilstein Online Database - Implementation, Content, and Retrieval; ACS Symposium Series #436, American Chemical Society, Washington, DC, 1990.
2. The Lycos internet address is: http://www.lycos.com.
3. The Alta-Vista internet address is: http://altavista.digital.com.
4. The InfoSeek internet address is: http://guide.infoseek.com.
5. The WebCrawler internet address is: http://webcrawler.com.
6. The Yahoo! Internet address is: http://www.yahoo.com.
7. Both the Beilstein Institute and Beilstein Information Systems are located at Varrentrappstrasse 40-42, Carl-Bosch Haus, Frankfurt (Main) 90, D-60486 Germany.
8. This figure is adapted from a talk by Bob Massie, CAS at the Herman Skolnik ACS Award symposium for Reiner Luckenbach and Clemens Jochum, Chicago, IL., August 1995.
9. Heller, S. R., Trends in Anal. Chem., 16, 1997, pp. 112-115 (1997). The Internet address for this article is: http://www.elsevier.nl:80/inca/homepage/saa/trac/frames.shtml